How many molecules are in a tablespoon?

Rachel Newton
Updated on April 27, 2026
How many molecules are in a tablespoon?
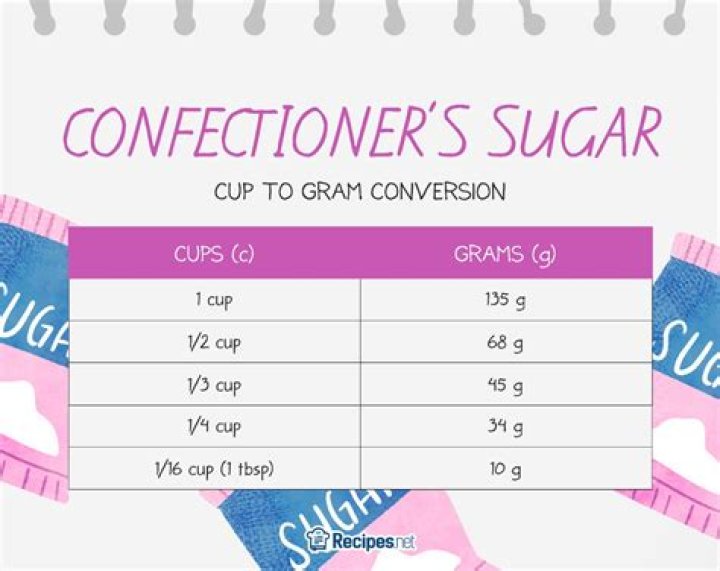
A US tablespoon of water will contain 4.943E23 molecules of water. A metric tablespoon will contain 5.014E23 molecules of water. An imperial tablespoon will contain 5.936E23 molecules of water.
How much can a tbsp hold?
In most places except Australia, one tablespoon equals three teaspoons—and one US tablespoon is 14.8 ml (0.50 US fl oz) or 15 ml (0.51 US fl oz).
How many moles are in a spoonful of water?
One mole of water is about 18 milliliters. This is the volume of a few drops of water, 3.65 teaspoons, 1.2 tablespoons, or 0.018 liters. It’s not a large volume, yet it contains 6.022 x 1023 water molecules! You can use the same steps to find the mass and volume of any other substance.
How many water molecules are in a teaspoon?
A teaspoon of water (about 5 mL) contains 2×10 23 water molecules, but each water molecule is comprised of 3 atoms: two hydrogen and one oxygen.
How many molecules are in a teaspoon of sugar?
A teaspoon of sugar is about 4.2 g. Table sugar is composed of sucrose, which is a disaccharide of glucose and fructose, with a molar mass of 342.3 g/mol. This means a sugar cube contains (4.2 / 342.3 =) 0.0123 mol of sucrose, or (0.0123 x 6.022 x 10^23 =) 7.39 x 10^21 molecules of sucrose.
What is the smallest molecule?
The smallest molecule is diatomic hydrogen (H2), with a bond length of 0.74 angstroms. Macromolecules are large molecules composed of smaller subunits; this term from biochemistry refers to nucleic acids, proteins, carbohydrates, and lipids.
Which has the greatest number of molecules?
Hence, 8.0 mole of H2 contains the largest number of molecules.
What is the mole of H2O?
The average mass of one H2O molecule is 18.02 amu. The number of atoms is an exact number, the number of mole is an exact number; they do not affect the number of significant figures. The average mass of one mole of H2O is 18.02 grams. This is stated: the molar mass of water is 18.02 g/mol.
How many molecules are in a tablespoon of water?
At 4 Celsius: A US tablespoon of water will contain 4.943E23 molecules of water. A metric tablespoon will contain 5.014E23 molecules of water. An imperial tablespoon will contain 5.936E23 molecules of water.
Do you know how many grams are in a tablespoon?
The reality is that both grams and tablespoons are two units of measurement that are very used in recipes. However, the reality is that when you are cooking or baking, you just want to know immediately the amount of an ingredient that you need to use.
Which is more a tablespoon or a teaspoon?
In nutrition labeling in both the United States and the United Kingdom, a tablespoon is defined as 15 ml. One tablespoon is exactly the same as 3 teaspoons as well as it is exactly the same as half fluid ounce. The tablespoon is currently used in three main countries: the United States, the United Kingdom, and in Australia.
How many ML is equal to a tablespoon?
But we will look into that further below. For now, what is important to keep in mind is that the United States Customary tablespoon is equal to approximately 14.8 l, a metric tablespoon is exactly equal to 15 ml, and an Australian tablespoon is equal to 20 ml.
At 4 Celsius: A US tablespoon of water will contain 4.943E23 molecules of water. A metric tablespoon will contain 5.014E23 molecules of water. An imperial tablespoon will contain 5.936E23 molecules of water.
The reality is that both grams and tablespoons are two units of measurement that are very used in recipes. However, the reality is that when you are cooking or baking, you just want to know immediately the amount of an ingredient that you need to use.
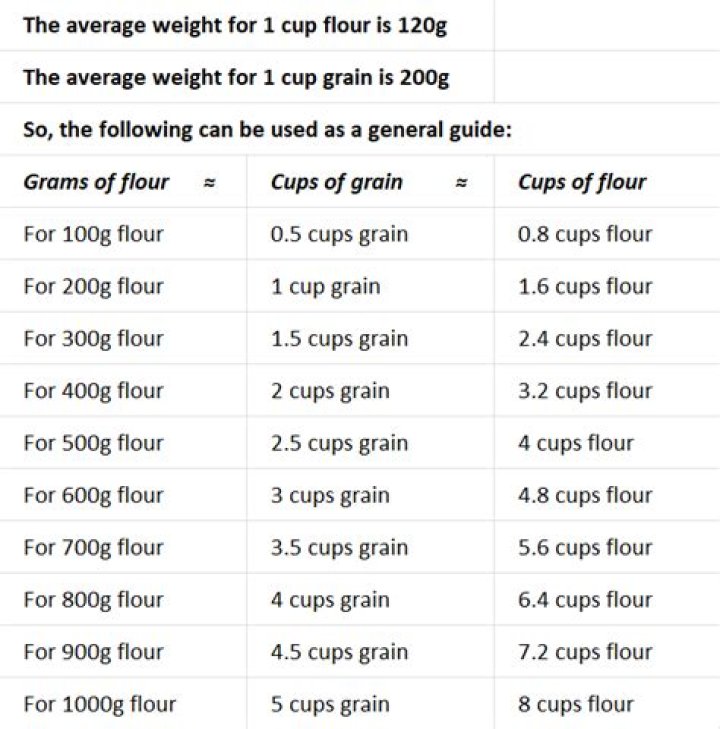
How to find how many milliliters are in a tablespoon?
To find how many milliliters [UK] are in a tablespoon, we divide the number of milliliters in a liter by the number of tablespoons in one liter. It can be expressed as; The Tablespoon to Milliliter conversion calculator is equipped with a blank text field and active controls that carry out independent functions of the converter.
How many molecules are in a gram of water?
Avogadro’s number for a gram-mol is 6.022E23 particles per mol. Hence the six answers: At 4 Celsius: A US tablespoon of water will contain 4.943E23 molecules of water. A metric tablespoon will contain 5.014E23 molecules of water.