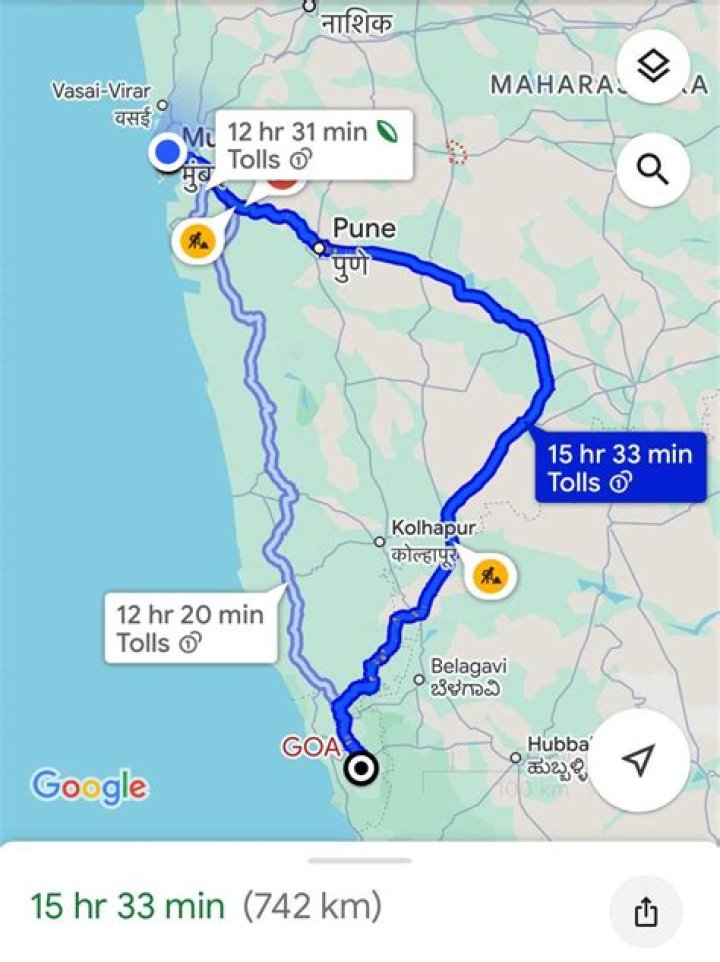
Is sodium hydrogen sulfate a hazardous material?

Rachel Newton
Updated on April 30, 2026
Is sodium hydrogen sulfate a hazardous material?
1 : Serious eye damage/eye irritation Category 1 H318 : Causes serious eye damage. R41 : Risk of serious damage to eyes.
What is the formula for sodium hydrogen sulfate?
NaHSO4
Sodium bisulfate/Formula
What is sodium hydrogen sulfate solution used for?
Sodium bisulfate is used primarily to lower pH. it also is used in metal finishing, cleaning products, and to lower the pH of water for effective chlorination in swimming pools and hot tubs.
Is sodium Hydrogensulfate corrosive?
Bisulfate, aqueous solution is a white crystalline solid dissolved in water. It is corrosive to metals and tissue.
What are the hazards of sodium sulfate?
Eye: May cause eye irritation. Causes redness and pain.
What should you do if you get brine solution on your hand?
Skin Contact: Remove contaminated clothing. Drench affected area with water for at least 15 minutes. Obtain medical attention if irritation develops or persists.
What is bisulfate of soda?
pHase® Bisulfate of Soda is a natural food acid that lowers pH without a sour taste. Products that benefit from Bisulfate of soda include sauces, dressings, soups, prepared meals, beverages, dips, fillings, snacks and desserts.
How do you make sodium bisulfate?
To prepare 10 mL of saturated sodium bisulfite:
- Add 4.75 g of sodium metabisulfite (di-sodium disulfite, Na2S2O5) to 6.25 mL of sterile, degassed ddH2O.
- Add 1.75 mL of 2 m NaOH.
- Add 1.25 mL of 1 m hydroquinone (0.11 g in 1 mL of H2O).
- Heat to 50°C in the dark, inverting the tube frequently.
- Adjust the pH to 5.0.
Is hydrogen sulfate the same as sulfuric acid?
The HSO4- ion is commonly referred to as hydrogen sulfate (which is also called the bisulfate ion). H2SO4 can be called dihydrogen sulfate (but it is popularly known as sulfuric acid).
Is sodium bisulfite bad for you?
► Sodium Bisulfite may cause mutations (genetic changes). Jersey Department of Health, Sodium Bisulfite has been tested and has not been shown to affect reproduction. low future exposure can cause itching and a skin rash. exposure may cause bronchitis to develop with coughing, phlegm, and/or shortness of breath.
What is the difference between sodium bisulfite and sodium bisulfate?
In inorganic chemistry|lang=en terms the difference between bisulfite and bisulfate. is that bisulfite is (inorganic chemistry) the univalent group -hso3, or any salt containing it while bisulfate is (inorganic chemistry) any salt containing this anion.
Can you drink sodium sulfate?
Do not swallow the dry powder. Shake well before use. Drink lots of noncaffeine liquids before, during, and after using sodium sulfate, potassium sulfate, and magnesium sulfate unless you are told to drink less liquid by your doctor.
A common ingredient in personal care products, sodium lauryl sulfate, or SLS, is an additive that allows cleansing products to foam. According to the Environmental Working Group’s Skin Deep Cosmetic Safety Database, SLS is a “moderate hazard” that has been linked to cancer, neurotoxicity, organ toxicity, skin irritation and endocrine disruption.
What is sodium hydrogen sulphate used for?
Sodium hydrogen sulfate can be used as an acidifier in foods as well as in the preparation of acidified sodium chlorite, an antimicrobial washing solution. The pKa values of various food acids are given below.
What is the formula for sodium hydrogen sulfite?
Sodium bisulfite (or sodium bisulphite, sodium hydrogen sulfite) is a chemical compound with the chemical formula NaHSO3. Sodium bisulfite is a food additive with E number E222. This salt of bisulfite can be prepared by bubbling sulfur dioxide in a solution of sodium carbonate in water.
Is sodium hydroxide and caustic soda the same?
Caustic soda, also known as sodium hydroxide and lye, is created during the same process of making chlorine. It is the principal strong base used in chemical applications. In most cases, the base is purchased as a 50% liquid solution. However, it is also available in dry forms such as pellets and granules.